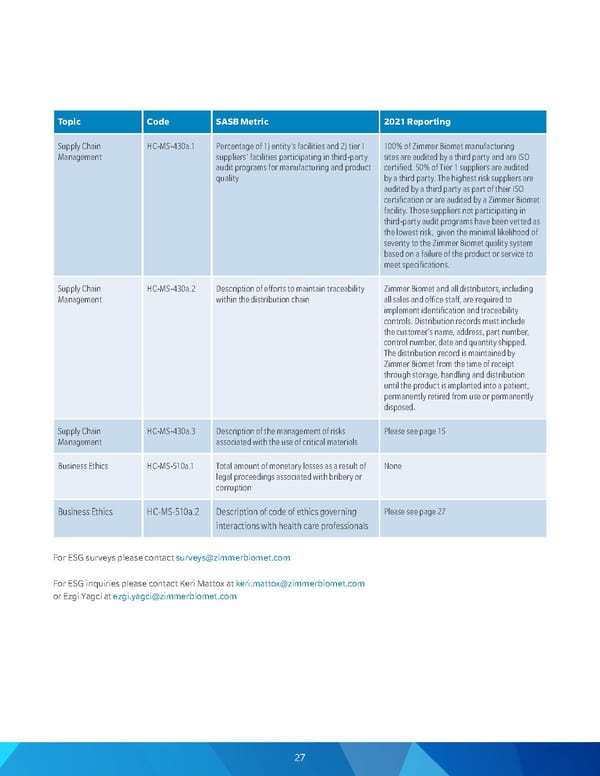
To p i c Code SASB Metric 2021 Reporting Supply Chain Management HC-MS-430a.1 Percentage of 1) entity's facilities and 2) tier I suppliers' facilities participating in third-party audit programs for manufacturing and product quality 100% of Zimmer Biomet manufacturing sites are audited by a third party and are ISO certified. 50% of Tier 1 suppliers are audited by a third party. The highest risk suppliers are audited by a third party as part of their ISO certification or are audited by a Zimmer Biomet facility. Those suppliers not participating in third-party audit programs have been vetted as the lowest risk, given the minimal likelihood of severity to the Zimmer Biomet quality system based on a failure of the product or service to meet specifications. Supply Chain Management HC-MS-430a.2 Description of efforts to maintain traceability within the distribution chain Zimmer Biomet and all distributors, including all sales and office staff, are required to implement identification and traceability controls. Distribution records must include the customer's name, address, part number, control number, date and quantity shipped. The distribution record is maintained by Zimmer Biomet from the time of receipt through storage, handling and distribution until the product is implanted into a patient, permanently retired from use or permanently disposed. Supply Chain Management HC-MS-430a.3 Description of the management of risks associated with the use of critical materials Please see page 15 Business Ethics H C - M S - 510 a .1 Total amount of monetary losses as a result of legal proceedings associated with bribery or corruption None Business Ethics HC-MS-510a.2 Description of code of ethics governing interactions with health care professionals Please see page 27 For ESG surveys please contact surveys@zimmerbiomet.com For ESG inquiries please contact Keri Mattox at keri.mattox@zimmerbiomet.com or Ezgi Yagci at ezgi.yagci@zimmerbiomet.com 27
 ZimmerBiomet Sustainability Report Page 28 Page 30
ZimmerBiomet Sustainability Report Page 28 Page 30